Scientists at TU Dresden’s Cluster of Excellence Physics of Life have identified a previously unknown mechanism by which large embryonic cells divide, directly contradicting a model that has appeared in biology textbooks for decades. The findings were published in Nature.
The established explanation for cell division centers on a contractile ring made of the protein actin. This ring tightens at a cell’s midpoint like a drawstring, pinching the cell into two daughter cells. The model works for most organisms, but it has never adequately explained division in species with exceptionally large embryonic cells, including sharks, birds, reptiles, and the platypus.
In those animals, the sheer volume of the cell and the presence of a large yolk sac physically prevent the actin ring from closing completely. For years, that left an open question: how do cells with a band that has loose ends generate enough force to split?
A Ratchet, Not a Ring
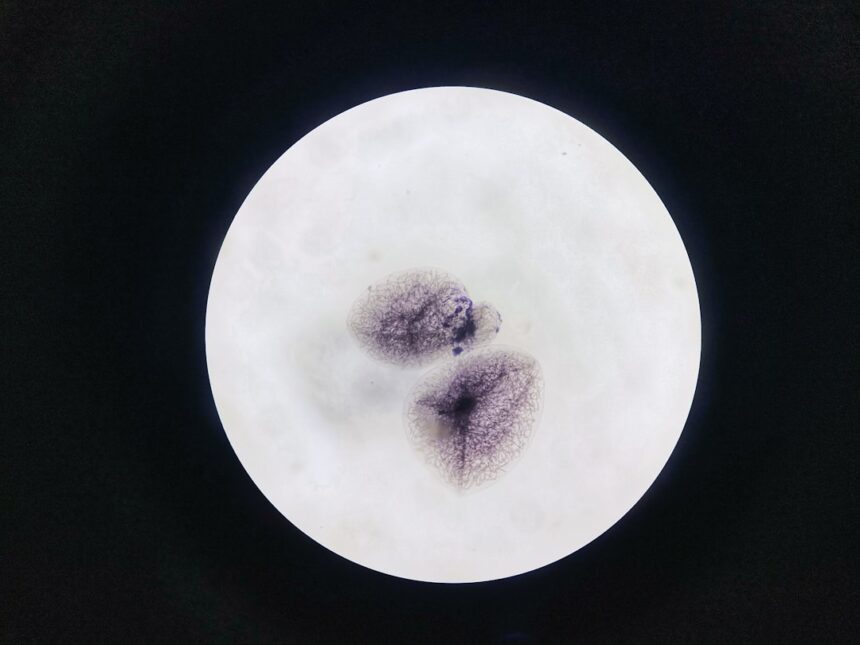
To find out, the Dresden team studied zebrafish embryos, which develop quickly and contain large, yolk-rich cells in their earliest stages. Lead author Alison Kickuth, a recently graduated PhD student from the Brugués group, used a laser to precisely sever the actin band during division. The band kept moving inward after being cut, indicating it was supported along its length rather than held only at its endpoints.
The researchers also noticed that microtubules, a separate component of the cell’s cytoskeleton, bent outward when the actin band was severed. This pointed to an active stabilizing role for microtubules during contraction.
To confirm that role, the team disrupted microtubules in two distinct ways: chemical depolymerization to stop new microtubules from forming, and physical interference by inserting a tiny oil droplet as an obstacle inside the cell. In both cases, the actin band collapsed. Without microtubule support, division failed.
The Cytoplasm Changes Stiffness on a Schedule
“With such a large yolk in the embryonic cell, there is a geometric constraint,” Kickuth said. “How does a contractile band, with loose ends, remain stable and generate enough force to divide these huge cells?”
The answer involves not just structure but timing. As cells move through the cell cycle, large microtubule formations called asters expand through the cytoplasm during interphase, the growth phase between divisions. Those asters appear to mark where the actin band will eventually form.
To measure whether the asters also change the mechanical properties of the cell interior, the team placed magnetic beads inside living cells and tracked their movement under an applied magnetic force. The measurements revealed that cytoplasmic stiffness changes predictably across different stages of the cell cycle. The asters, by influencing stiffness, help anchor the actin band as it contracts.
What the Model Means
Together, the findings describe what the researchers call a “mechanical ratchet” system. Rather than one continuous tightening motion, the actin band advances in incremental steps, held in place between each contraction by cytoskeletal support and rhythmic shifts in the cytoplasm’s physical properties.
- Microtubules stabilize the actin band along its length, not just at its ends
- Cytoplasmic stiffness oscillates with the cell cycle, creating anchor points
- The system allows division without a fully closed contractile ring
The mechanism applies specifically to large, yolk-bearing cells but raises broader questions about whether similar dynamics operate in other contexts where the standard purse-string model has been assumed to hold.
Photo by Cheng Qi Huang on Unsplash
This article is a curated summary based on third-party sources. Source: Read the original article